- About Us
- Services & Solutions
- Novel Technology Platform
- Bioconjugation Discovery
- Bioconjugation Process Development
- Analytical Development
- Formulation and DP Development
- GMP mAb Intermediate and Conjugation Manufacturing
- GMP Formulation, Fill and Lyophilization
- Payload and Linker R&D and Manufacturing
- Ready-Made Payloads & Linkers
- WuXi XDC Singapore Site
- Investor Relations
- News & Resources
WuXi Biologics
Offering End-to-End Solutions
Expert Blog
Accelerating ADC Discovery Through Integrated Expertise

By Dr. Gang Yin
Vice President, Head of Discovery Service Team
In the rapidly evolving field of oncology and targeted therapeutics, antibody–drug conjugates (ADCs) represent one of the most sophisticated and promising modalities, combining the specificity of monoclonal antibodies with the potency of cytotoxic or non-cytotoxic payloads. However, developing an effective ADC is a complex, multi-step process—spanning antibody selection, payload optimization, linker chemistry, conjugation, purification, and extensive analytical characterization, as well as in vitro assessments such as antigen binding, internalization, and cytotoxicity, and in vivo studies covering pharmacokinetics (PK), pharmacodynamics (PD), tumor inhibition, and safety.
Establishing and managing all of these functions in-house is costly and time-consuming. Working with multiple CROs introduces additional challenges, including extensive coordination, import/export logistics, hand-offs between vendors, and increased risks of delays or loss of accountability. Consequently, identifying a lead preclinical candidate often takes over 24 months and requires significant investment.
Integrating all discovery functions within a single platform is critical. This approach ensures seamless workflows, accelerates decision-making, reduces development bottlenecks, and significantly shortens both costs and timelines from concept to candidate selection. WuXi XDC, a global leader in integrated ADC discovery, combines end-to-end, one-stop discovery services, world-class scientists, advanced labs and equipment, and extensive ADC experience to accelerate the timeline from target identification to fully characterized ADC candidates to 8–10 months, enabling faster, more efficient innovation for our partners.
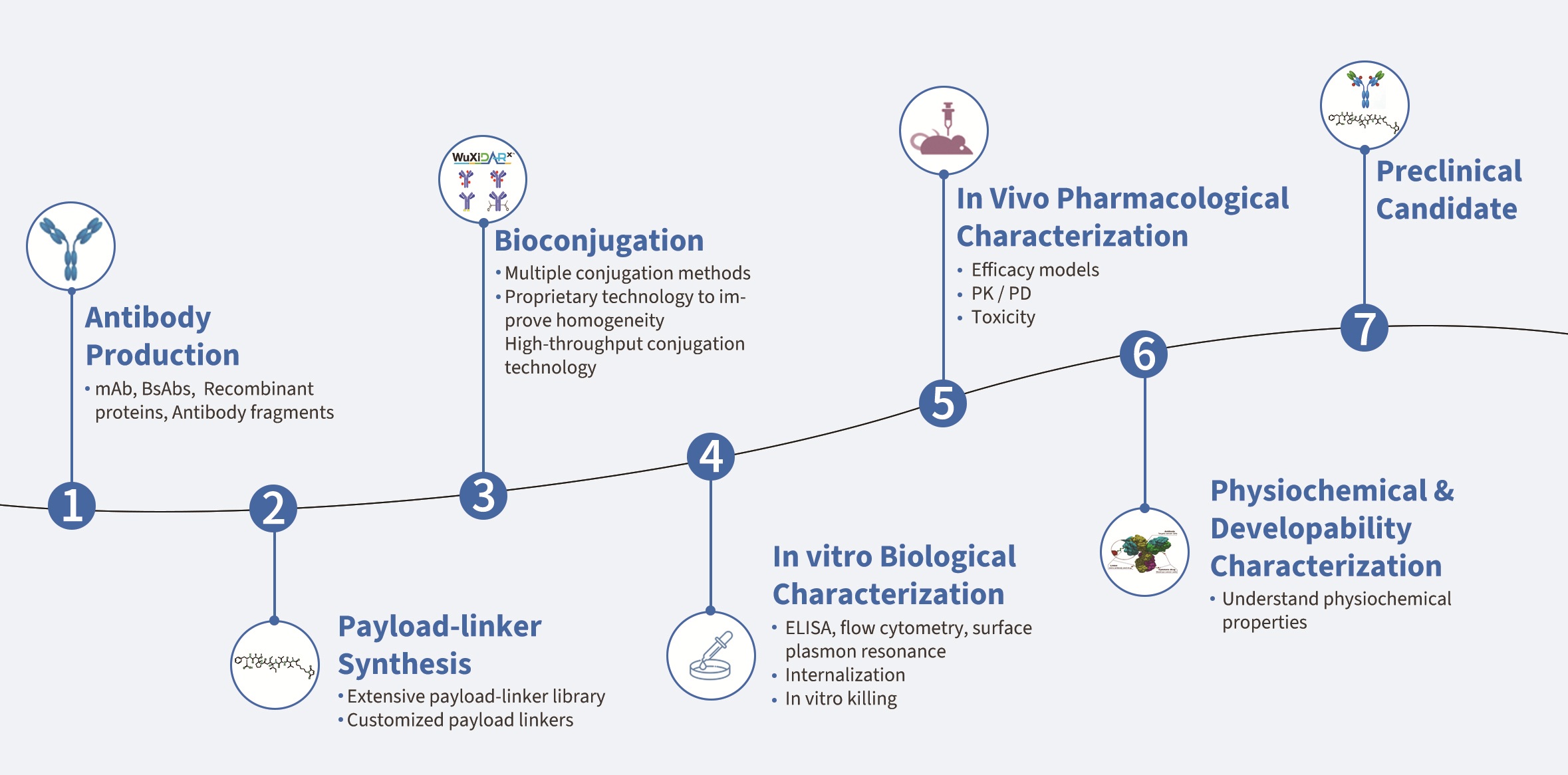
ADC lead selection timeline: ~ 10 months for one round through integrated discovery platform
- Highlights of Bioconjugation Discovery Service
WuXi XDC’s bioconjugation discovery platform is supported by a team of over 150 highly trained and talented scientists in state-of-the art labs. Beyond its bioconjugation teams, WuXi XDC Discovery Service has a dedicated ADC purification group equipped with more than 40 state-of-the-art, high-end instruments, along with a specialized analytical team operating over 30 cutting-edge, industry-leading analytical instruments. The company is continuously expanding its lab space and growing its team. By the end of 2025, WuXi XDC had successfully delivered over 19,000 conjugates across more than 900 projects for over 500 clients worldwide.

WuXi XDC bioconjugation discovery service highlight
The Bioconjugation Discovery team possesses extensive expertise in a broad range of conjugation methods, including interchain cysteine, engineered cysteine, lysine, glycan, and enzyme-mediated conjugations. Additionally, WuXi XDC developed the proprietary WuXiDARx™ conjugation platform, the only site-specific, homogeneous conjugation technology based on interchain cysteines, which enables precise control of drug–antibody ratio (DAR) and improves ADC consistency and safety.
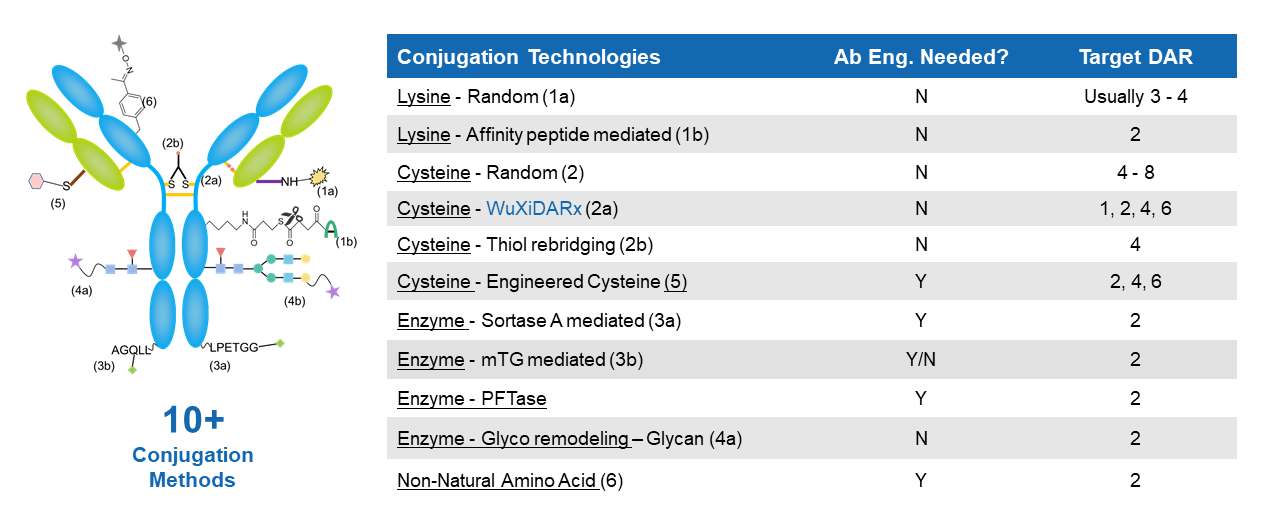
Extensive expertise in a broad range of conjugation methods
While mAb IgG1 remains the most commonly used ADC carrier, WuXi XDC has enabled conjugation across a wide variety of scaffolds, including VHH, VHH-Fc, Fab, over a dozen bispecific antibody (BsAb) formats, and other innovative protein scaffolds, providing flexibility to design ADCs tailored to diverse therapeutic needs.
The dedicated ADC purification team has extensive experience in process design and optimization for column purification and ultrafiltration/diafiltration. The team has implemented an automated purification platform capable of handling projects ranging from milligram to 100-gram scale to support drug discovery. In parallel, the analytical team operates a comprehensive Bioconjugate Analytical Panel, including concentration, purity, DAR, free drug, endotoxin, charge variants, and modifications. To further support ADC development, the analytical team offers mini-stability and extensive developability studies, evaluating freeze/thaw stability, short- and long-term storage, thermal stress, light sensitivity, concentratability, and other critical attributes.
- Highlights of Payload-Linker Discovery Service
WuXi XDC’s Payload-Linker Discovery team brings industry-leading expertise across a wide variety of payload and linker classes, including commonly used cytotoxic and non-cytotoxic payloads, cleavable and non-cleavable linkers, and fully customized payload–linker combinations designed to meet specific client needs. The team provides chemical synthesis services and maintains an extensive payload–linker library, offering clients a broad selection of ready-to-use building blocks. For projects requiring unique therapeutic strategies, WuXi XDC offers custom synthesis of linker-payloads tailored to precise specifications, enabling rapid exploration of novel ADC designs. This integrated approach accelerates the identification of optimal payload–linker combinations, reduces development timelines, and enhances ADC efficacy and safety profiles.
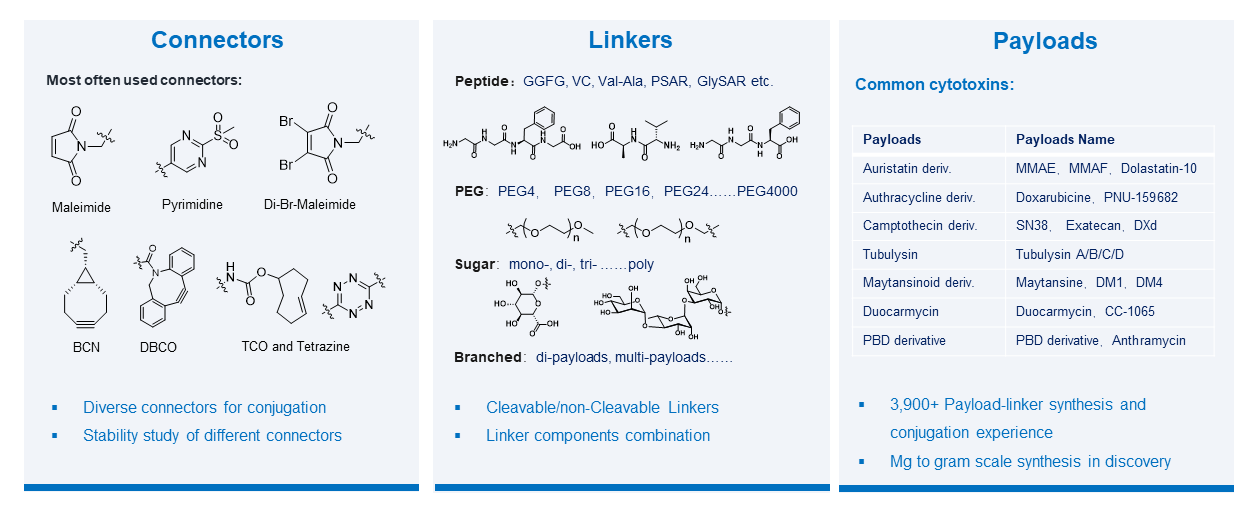
Deep Expertise across a wide variety of payload and linker classes
Payload-linker discovery service team has extensive experience with diverse mechanisms of action (MOAs) for non-cytotoxic payloads as well, including degraders for degrader-antibody conjugates (DACs), oligonucleotides for antibody-oligonucleotide conjugates (AOCs), peptides for antibody-peptide conjugates (APCs), immune agonists, DNA damage response inhibitors (DDRis), and others. These capabilities expand the scope of payload options for novel ADC discovery and development. In addition, the platform offers flexible combination strategies to support dual-payload ADC development.
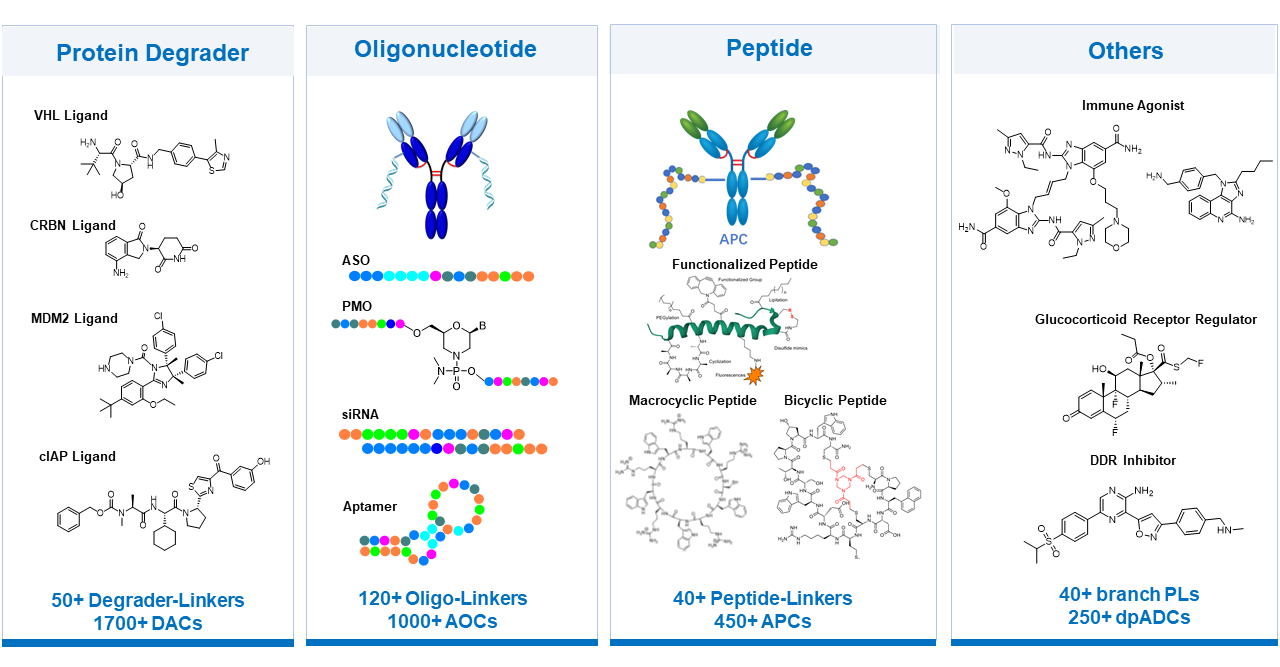
Extensive Experience with Diverse MOAs of Payloads at WuXi XDC: Flexible in Combination Strategies
- WuXi XDC’s Proprietary Innovations
WuXi XDC’s proprietary innovations include the WuXiDARx™ conjugation platform, X-LinC linker, and WuXiTecan linker-payload.
- WuXiDARx™ is the only site-specific conjugation platform based on inter-chain cysteines, eliminating the need for antibody engineering, enzymes, or column purification. It enables the generation of DAR1, DAR2, DAR4, and DAR6 site-specific, homogeneous conjugates, providing precise control over drug–antibody ratio and improving ADC consistency.
- X-LinC is a novel linker that demonstrates superior stability compared with traditional maleimide linkers, translating to improved safety profiles and enhanced in vivo performance.
- WuXiTecan-2™ is a proprietary linker–payload designed to maintain comparable efficacy while reducing toxicity, offering a safer therapeutic window for ADC development.
These platforms collectively illustrate WuXi XDC’s commitment to advancing ADC design, providing clients with highly precise, stable, and safe conjugation options to accelerate discovery and development.

WuXi XDC’s Proprietary Technology Platforms(WuXiDARxTM is in collaboration with MCLICK-DAR1-A1/MCLICK-DAR2-A1/MCLICK-DAR6-A1 from Bio-reinnovation Tech. Ltd.)
- Extensive Experience in Bioconjugate Discovery
In 2025, WuXi XDC explored a remarkable total of over 5,600 bioconjugate molecules, underscoring our extensive experience and leadership in the field. Among these, approximately 3,400 were ADC molecules, with the majority (~2,800) being traditional ADCs, alongside a growing number of novel formats such as bispecific ADCs (~360) and dual-payload ADCs (~230). This reflects the industry’s ongoing push toward more complex and multifunctional antibody therapeutics designed to improve efficacy and overcome resistance mechanisms.
In parallel, nearly 1,700 novel bioconjugate molecules were developed, including degrader antibody conjugates (DACs, ~530), antibody oligonucleotide conjugates (AOCs, ~350), antibody peptide conjugates (APCs, ~190), and others, demonstrating the expanding landscape beyond classic ADCs into next-generation conjugates designed to target a broader range of diseases and cellular pathways.
This extensive molecule portfolio and diversification highlight WuXi XDC’s deep expertise and agility in bioconjugate discovery, enabling the company to stay at the forefront of evolving therapeutic trends and deliver cutting-edge solutions that meet the demands of modern drug development.

Over 5,600 ADC/XDC Molecules Explored at WuXi XDC in 2025
With talented teams, proprietary platforms, and deep ADC expertise, WuXi XDC accelerates discovery from concept to fully characterized candidates. We aim to help partners bring next-generation antibody therapies to patients faster and more efficiently.
Abbreviation:
ELISA=enzyme-linked immunosorbent assay
PK=pharmacokinetics
PD=Pharmacodynamics
AOC=Antibody Oligonucleotide Conjugate
DAC=Degrader Antibody Drug Conjugate
APC=Antibody Peptide Conjugate
CDX = Cell line-derived xenograft model
About the Author
Dr. Gang Yin is Head of Discovery Services at WuXi XDC. He leads global efforts to deliver integrated discovery solutions for ADCs and next-generation bioconjugates.
Boasting over 20 years of industry experience, he is a seasoned expert in ADC discovery and site-specific conjugation technologies, he has extensive experience in bioconjugation, linker–payload design, and deep expertise in advancing complex biologics from early-stage discovery through preclinical development and IND-enabling studies in preparation for clinical trials.
He has made significant contributions to six ADC programs—including bispecific and dual-payload ADCs—as well as vaccine and cytokine conjugates, all of which advanced into clinical development. He is an author of numerous publications and patents in the field of ADCs.
Related Resources
Notice:
You are leaving WuXi Biologics Website, after which our Privacy Notice will not apply. Please keep it in mind the protection of your privacy. Are you willing to proceed?









