- About Us
- Services & Solutions
- Novel Technology Platform
- Bioconjugation Discovery
- Bioconjugation Process Development
- Analytical Development
- Formulation and DP Development
- GMP mAb Intermediate and Conjugation Manufacturing
- GMP Formulation, Fill and Lyophilization
- Payload and Linker R&D and Manufacturing
- Ready-Made Payloads & Linkers
- WuXi XDC Singapore Site
- Investor Relations
- News & Resources
WuXi Biologics
Offering End-to-End Solutions
Services & Solutions
Vaccine Product Development & Manufacturing
WuXi Vaccines is an Integrated Global CDMO Platform for Vaccine Development and Manufacturing. With leading technical platforms, including mammalian, microbial, VLP, viral and mRNA, we ensure quick clinical and market access to vaccine products. Committed to the global collaboration that enables our partners in development and manufacturing of vaccine products, we are proud to support small start-ups and multinational corporations alike in bringing more products to the market, more efficiently, to advance and benefit global health initiatives.
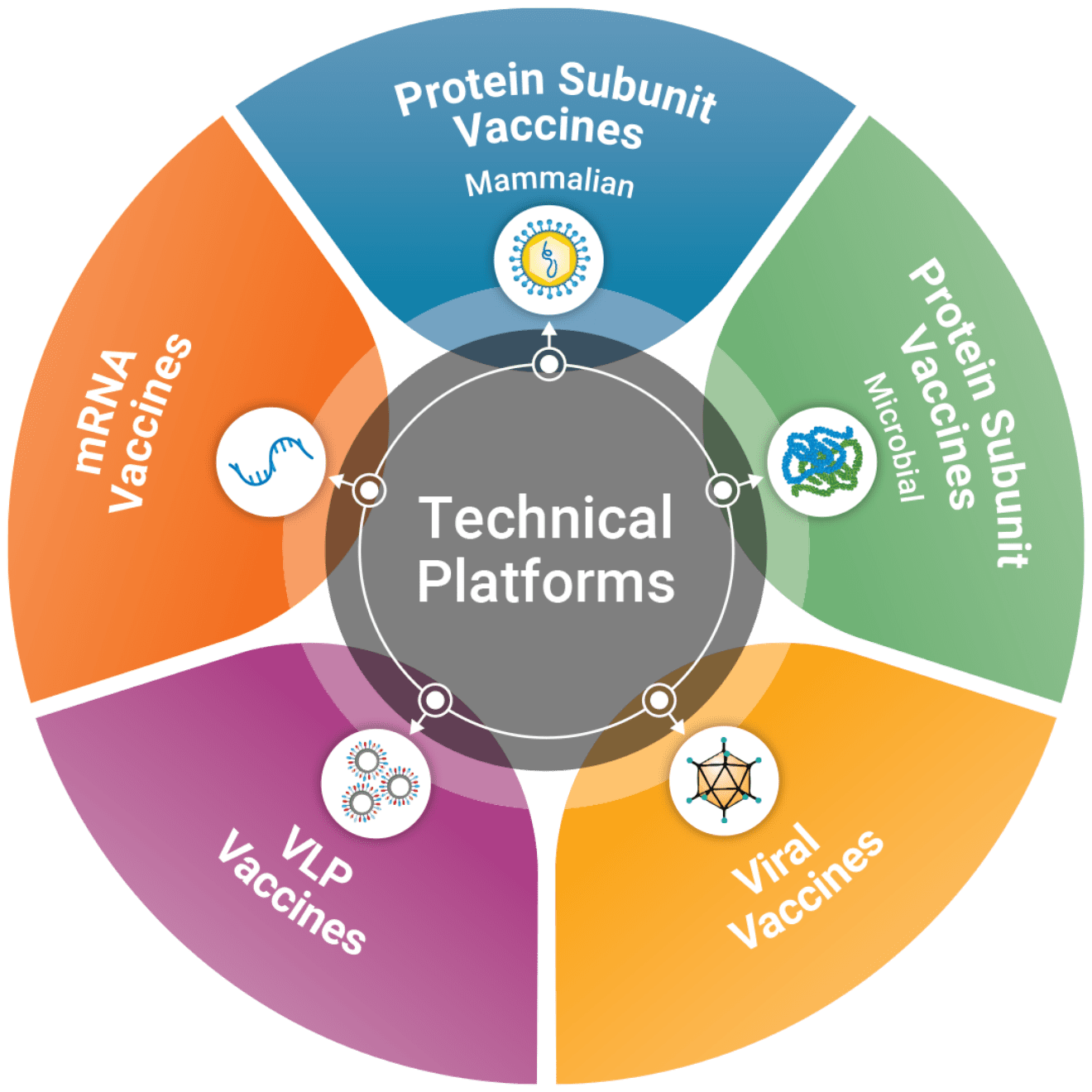
With the experience and technology to develop and manufacture many types of vaccines including viral vector, live attenuated virus, DNA, mRNA/RNA, VLP, and recombinant protein subunit vaccines, we strive to enable global partners to deliver critical vaccines anywhere in the world. Regardless of vaccine modality, we leverage our technical expertise, broad regulatory knowledge, premium quality system, advanced CMC development capabilities, multiple production platforms (mammalian, microbial) and extensive GMP manufacturing capacities to provide a true end-to-end service from vaccine development to large-scale commercial production and distribution.


End-to-End Capabilities
Using our comprehensive testing and manufacturing services, we support the development of vaccine products at any stage. From initial vector screening or protein generation to late stage manufacturing, we offer the entire range of support services such as developability assessment, cell line development & banking, analytical development, process development, drug product (DP) formulation, drug substance and drug product manufacturing, and quality control lot release.

Notice:
You are leaving WuXi Biologics Website, after which our Privacy Notice will not apply. Please keep it in mind the protection of your privacy. Are you willing to proceed?





